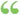 F,o. 19. always more nearly horizontal than the former, as has been proved both experimentally and theoretically. The freezing-point of a liquid is that temperature at which the solid and liquid states can exist side by side indefinitely. This condition... F,o. 19. always more nearly horizontal than the former, as has been proved both experimentally and theoretically. The freezing-point of a liquid is that temperature at which the solid and liquid states can exist side by side indefinitely. This condition...  A Text-book of Inorganic Chemistry - Page 61by Arnold Frederik Holleman - 1902 - 458 pagesFull view A Text-book of Inorganic Chemistry - Page 61by Arnold Frederik Holleman - 1902 - 458 pagesFull view - About this book
 | Arnold Frederik Holleman - Chemistry, Inorganic - 1911 - 522 pages
...gives the pressures for the frozen matter; the part be for the liquid solvent. This latter part is always more nearly horizontal than the former, as...from the solid condensing to a liquid and the former grad71 Fio. 18. Fio. 19. ually turning into the latter. Inversely, if the vapor tension of the solid... | |
 | Arthur Amos Noyes - Chemistry, Physical and theoretical - 1922 - 360 pages
...FREEZING-POINT OF SOLUTIONS 48. Freezing-Point and Its Relation to Vapor-Pressure and Molal Composition. — The freezing-point of a liquid is that temperature at which the solid solvent and the liquid coexist in equilibrium with each other. The solid which separates from the solution... | |
| |